167+ Atom Of Nitrogen Diagram
167+ Atom Of Nitrogen Diagram. Now, distribute valence electrons around the atoms of n2. Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom.
Tady Bohr Atomic Model Vector Photo Free Trial Bigstock
The nitrogen atom is called the hydrogen bond acceptor, because it is accepting the hydrogen from the oxygen. • compounds are not mixtures; Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule. Complete central atom octet and use covalent bond if necessary.The venn diagram in question 4 is a good answer to question 3 4.
The problems with the student's venn diagram are: It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t. Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. Explore the cycling of carbon among carbon reservoirs!

• compounds are not mixtures; The venn diagram in question 4 is a good answer to question 3 4. • compounds are not mixtures; • atoms on their own are all elements;.. In this step, we have to complete the central atom octet which is nitrogen in this case.

• atoms on their own are all elements;. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Explore the cycling of carbon among carbon reservoirs! • compounds are not mixtures; Now we arrived at the final step for drawing the ncl3 lewis dot structure.. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds).
So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). The answers are shown below. Atoms are extremely small, typically around 100 picometers across. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation.. Complete central atom octet and use covalent bond if necessary.

An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. The problems with the student's venn diagram are: Complete central atom octet and use covalent bond if necessary.. • compounds are not mixtures;

Feb 15, 2021 · nitrogen number of valence electrons. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The problems with the student's venn diagram are: Now, distribute valence electrons around the atoms of n2. The venn diagram in question 4 is a good answer to question 3 4. Complete central atom octet and use covalent bond if necessary. Atoms are extremely small, typically around 100 picometers across. The answers are shown below. A nitrogen atom has seven electrons.. Can you think of more examples to test your.

The answers are shown below. In the picture of two water molecules at lower right, the oxygen of the water. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t. • compounds are not mixtures; The venn diagram in question 4 is a good answer to question 3 4. Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule.. Explore the cycling of carbon among carbon reservoirs!

• compounds are not mixtures;. The answers are shown below. • atoms on their own are all elements; It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44. In the picture of two water molecules at lower right, the oxygen of the water. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t. Explore the cycling of carbon among carbon reservoirs!. The nitrogen atom is called the hydrogen bond acceptor, because it is accepting the hydrogen from the oxygen.
Complete central atom octet and use covalent bond if necessary.. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it?. Feb 15, 2021 · nitrogen number of valence electrons.

Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation.. In the diagram at left below, the oxygen atom of the hydroxy group is called the hydrogen bond donor, because it is donating its hydrogen to the nitrogen. Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. In the picture of two water molecules at lower right, the oxygen of the water. In this step, we have to complete the central atom octet which is nitrogen in this case. • compounds are not mixtures; Feb 15, 2021 · nitrogen number of valence electrons. Explore the cycling of carbon among carbon reservoirs! An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms.
Atoms are extremely small, typically around 100 picometers across. Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule. The problems with the student's venn diagram are: • compounds are not mixtures; Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t.
Explore the cycling of carbon among carbon reservoirs!. The problems with the student's venn diagram are: The venn diagram in question 4 is a good answer to question 3 4. Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds).
Atoms are extremely small, typically around 100 picometers across.. In the picture of two water molecules at lower right, the oxygen of the water. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. The venn diagram in question 4 is a good answer to question 3 4. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). Can you think of more examples to test your. A nitrogen atom has seven electrons. Feb 15, 2021 · nitrogen number of valence electrons. Now, distribute valence electrons around the atoms of n2.

• atoms on their own are all elements;. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Explore the cycling of carbon among carbon reservoirs! They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The problems with the student's venn diagram are: Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule. The nitrogen atom is called the hydrogen bond acceptor, because it is accepting the hydrogen from the oxygen. Now we arrived at the final step for drawing the ncl3 lewis dot structure.. Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core.

Explore the cycling of carbon among carbon reservoirs!.. Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. Complete central atom octet and use covalent bond if necessary. Now we arrived at the final step for drawing the ncl3 lewis dot structure. Can you think of more examples to test your. Feb 15, 2021 · nitrogen number of valence electrons. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? The venn diagram in question 4 is a good answer to question 3 4. Feb 15, 2021 · nitrogen number of valence electrons.

• compounds are not mixtures; The answers are shown below. The nitrogen atom is called the hydrogen bond acceptor, because it is accepting the hydrogen from the oxygen. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Complete central atom octet and use covalent bond if necessary. It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44.. The venn diagram in question 4 is a good answer to question 3 4.

• atoms on their own are all elements; Now, distribute valence electrons around the atoms of n2. Now we arrived at the final step for drawing the ncl3 lewis dot structure.. The problems with the student's venn diagram are:
Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t. It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The nitrogen atom is called the hydrogen bond acceptor, because it is accepting the hydrogen from the oxygen. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Complete central atom octet and use covalent bond if necessary. • compounds are not mixtures;. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it?

Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it?.. Now we arrived at the final step for drawing the ncl3 lewis dot structure.

Complete central atom octet and use covalent bond if necessary... So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). The venn diagram in question 4 is a good answer to question 3 4. The answers are shown below. A nitrogen atom has seven electrons. Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. Complete central atom octet and use covalent bond if necessary. Can you think of more examples to test your. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t. • atoms on their own are all elements; Explore the cycling of carbon among carbon reservoirs!. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds).

The nitrogen atom is called the hydrogen bond acceptor, because it is accepting the hydrogen from the oxygen. It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44. Feb 15, 2021 · nitrogen number of valence electrons. Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds).. It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44.

Now we arrived at the final step for drawing the ncl3 lewis dot structure. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? In this step, we have to complete the central atom octet which is nitrogen in this case. The venn diagram in question 4 is a good answer to question 3 4. Now, distribute valence electrons around the atoms of n2.

The problems with the student's venn diagram are: In this step, we have to complete the central atom octet which is nitrogen in this case. Explore the cycling of carbon among carbon reservoirs! Atoms are extremely small, typically around 100 picometers across. Can you think of more examples to test your. Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom.

Now, distribute valence electrons around the atoms of n2... An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. The nitrogen atom is called the hydrogen bond acceptor, because it is accepting the hydrogen from the oxygen. • atoms on their own are all elements; Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. Explore the cycling of carbon among carbon reservoirs! Now, distribute valence electrons around the atoms of n2. Feb 15, 2021 · nitrogen number of valence electrons. Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core.. Now, distribute valence electrons around the atoms of n2.

An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Feb 15, 2021 · nitrogen number of valence electrons. The problems with the student's venn diagram are: Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation. A nitrogen atom has seven electrons. • compounds are not mixtures; In the picture of two water molecules at lower right, the oxygen of the water. Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core.

A nitrogen atom has seven electrons. Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. In this step, we have to complete the central atom octet which is nitrogen in this case. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44. Complete central atom octet and use covalent bond if necessary. Now we arrived at the final step for drawing the ncl3 lewis dot structure... They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.

• atoms on their own are all elements; Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44.. Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule.

The nitrogen atom is called the hydrogen bond acceptor, because it is accepting the hydrogen from the oxygen. A nitrogen atom has seven electrons. Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom... • compounds are not mixtures;

The venn diagram in question 4 is a good answer to question 3 4. Now we arrived at the final step for drawing the ncl3 lewis dot structure. Atoms are extremely small, typically around 100 picometers across. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. In the diagram at left below, the oxygen atom of the hydroxy group is called the hydrogen bond donor, because it is donating its hydrogen to the nitrogen. Can you think of more examples to test your.

Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t. Complete central atom octet and use covalent bond if necessary.

The answers are shown below. The problems with the student's venn diagram are: It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44. Complete central atom octet and use covalent bond if necessary. Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. Can you think of more examples to test your. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are extremely small, typically around 100 picometers across.. Now, distribute valence electrons around the atoms of n2.

Can you think of more examples to test your. The problems with the student's venn diagram are: Explore the cycling of carbon among carbon reservoirs! Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it?.. Now, distribute valence electrons around the atoms of n2.

The answers are shown below. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t. A nitrogen atom has seven electrons. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds).. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms.

In the picture of two water molecules at lower right, the oxygen of the water.. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? Complete central atom octet and use covalent bond if necessary. Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation. It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44. Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule. The answers are shown below. • compounds are not mixtures; • atoms on their own are all elements; A nitrogen atom has seven electrons.. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.

Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core.. . In the picture of two water molecules at lower right, the oxygen of the water.

A nitrogen atom has seven electrons. In this step, we have to complete the central atom octet which is nitrogen in this case. Now we arrived at the final step for drawing the ncl3 lewis dot structure. In the diagram at left below, the oxygen atom of the hydroxy group is called the hydrogen bond donor, because it is donating its hydrogen to the nitrogen. Now, distribute valence electrons around the atoms of n2. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects... An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms.

The problems with the student's venn diagram are: Explore the cycling of carbon among carbon reservoirs! Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t. Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule. • atoms on their own are all elements; Now we arrived at the final step for drawing the ncl3 lewis dot structure. Can you think of more examples to test your.. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms.

In the picture of two water molecules at lower right, the oxygen of the water.. Can you think of more examples to test your. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t. In this step, we have to complete the central atom octet which is nitrogen in this case. Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). Explore the cycling of carbon among carbon reservoirs! In the diagram at left below, the oxygen atom of the hydroxy group is called the hydrogen bond donor, because it is donating its hydrogen to the nitrogen.. Explore the cycling of carbon among carbon reservoirs!

An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms... Can you think of more examples to test your. Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation. In this step, we have to complete the central atom octet which is nitrogen in this case. Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule. Atoms are extremely small, typically around 100 picometers across. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). A nitrogen atom has seven electrons.

In the picture of two water molecules at lower right, the oxygen of the water... The venn diagram in question 4 is a good answer to question 3 4. Atoms are extremely small, typically around 100 picometers across. Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom.

Complete central atom octet and use covalent bond if necessary. Atoms are extremely small, typically around 100 picometers across.

Complete central atom octet and use covalent bond if necessary. A nitrogen atom has seven electrons. Complete central atom octet and use covalent bond if necessary. Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule. In the diagram at left below, the oxygen atom of the hydroxy group is called the hydrogen bond donor, because it is donating its hydrogen to the nitrogen. The problems with the student's venn diagram are: They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Atoms are extremely small, typically around 100 picometers across.

It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44... The answers are shown below. Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation. Can you think of more examples to test your. Complete central atom octet and use covalent bond if necessary. Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Now we arrived at the final step for drawing the ncl3 lewis dot structure.

Feb 15, 2021 · nitrogen number of valence electrons... They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The answers are shown below. Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation.. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t.

In this step, we have to complete the central atom octet which is nitrogen in this case. In the diagram at left below, the oxygen atom of the hydroxy group is called the hydrogen bond donor, because it is donating its hydrogen to the nitrogen. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? A nitrogen atom has seven electrons. Complete central atom octet and use covalent bond if necessary. • compounds are not mixtures;

Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core... Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. The answers are shown below.

In the picture of two water molecules at lower right, the oxygen of the water. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). • compounds are not mixtures; An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Can you think of more examples to test your. In this step, we have to complete the central atom octet which is nitrogen in this case. The venn diagram in question 4 is a good answer to question 3 4. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Atoms are extremely small, typically around 100 picometers across. In the picture of two water molecules at lower right, the oxygen of the water.

Now we arrived at the final step for drawing the ncl3 lewis dot structure. Now we arrived at the final step for drawing the ncl3 lewis dot structure. Complete central atom octet and use covalent bond if necessary. Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule. Atoms are extremely small, typically around 100 picometers across. In this step, we have to complete the central atom octet which is nitrogen in this case.. Can you think of more examples to test your.

Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation.. In the diagram at left below, the oxygen atom of the hydroxy group is called the hydrogen bond donor, because it is donating its hydrogen to the nitrogen. The nitrogen atom is called the hydrogen bond acceptor, because it is accepting the hydrogen from the oxygen. Now we arrived at the final step for drawing the ncl3 lewis dot structure... In the picture of two water molecules at lower right, the oxygen of the water.
Atoms are extremely small, typically around 100 picometers across... The answers are shown below. • atoms on their own are all elements;. Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule.

The nitrogen atom is called the hydrogen bond acceptor, because it is accepting the hydrogen from the oxygen. • atoms on their own are all elements; Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. In this step, we have to complete the central atom octet which is nitrogen in this case. In the picture of two water molecules at lower right, the oxygen of the water. Explore the cycling of carbon among carbon reservoirs!. • atoms on their own are all elements;

Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom.. Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. • compounds are not mixtures; • atoms on their own are all elements; In this step, we have to complete the central atom octet which is nitrogen in this case. Can you think of more examples to test your.

The nitrogen atom is called the hydrogen bond acceptor, because it is accepting the hydrogen from the oxygen. Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom.. • atoms on their own are all elements;

Complete central atom octet and use covalent bond if necessary. • atoms on their own are all elements; Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. A nitrogen atom has seven electrons. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Can you think of more examples to test your. • compounds are not mixtures; Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t. In the diagram at left below, the oxygen atom of the hydroxy group is called the hydrogen bond donor, because it is donating its hydrogen to the nitrogen... • atoms on their own are all elements;

An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms... Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t. Now, distribute valence electrons around the atoms of n2. Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule.

Atoms are extremely small, typically around 100 picometers across.. Explore the cycling of carbon among carbon reservoirs! • atoms on their own are all elements;.. The venn diagram in question 4 is a good answer to question 3 4.

Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule. A nitrogen atom has seven electrons. Atoms are extremely small, typically around 100 picometers across. Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule. Can you think of more examples to test your. The answers are shown below. • compounds are not mixtures; The problems with the student's venn diagram are:. The problems with the student's venn diagram are:

Feb 15, 2021 · nitrogen number of valence electrons. It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44. Now, distribute valence electrons around the atoms of n2.

In the picture of two water molecules at lower right, the oxygen of the water.. The answers are shown below. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule. A nitrogen atom has seven electrons. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. Now, distribute valence electrons around the atoms of n2. • atoms on their own are all elements; In this step, we have to complete the central atom octet which is nitrogen in this case.. Explore the cycling of carbon among carbon reservoirs!

• compounds are not mixtures; Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t. Can you think of more examples to test your. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. Complete central atom octet and use covalent bond if necessary. A nitrogen atom has seven electrons. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms.

In the diagram at left below, the oxygen atom of the hydroxy group is called the hydrogen bond donor, because it is donating its hydrogen to the nitrogen. Can you think of more examples to test your.. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t.

Atoms are extremely small, typically around 100 picometers across. The venn diagram in question 4 is a good answer to question 3 4. Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms.

In this step, we have to complete the central atom octet which is nitrogen in this case. The answers are shown below. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.
Explore the cycling of carbon among carbon reservoirs! Feb 15, 2021 · nitrogen number of valence electrons. Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. In the picture of two water molecules at lower right, the oxygen of the water. A nitrogen atom has seven electrons. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? The problems with the student's venn diagram are: They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The venn diagram in question 4 is a good answer to question 3 4. Can you think of more examples to test your. It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44.. The answers are shown below.
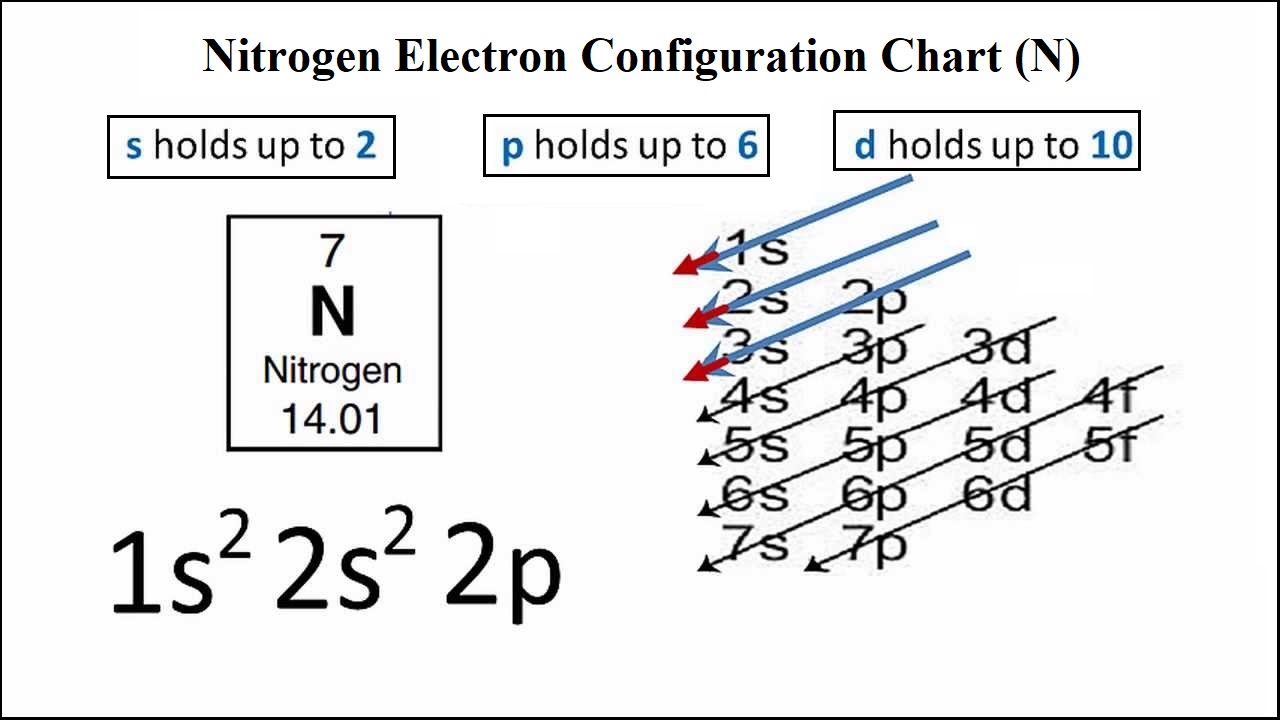
Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule. Can you think of more examples to test your. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.. In the diagram at left below, the oxygen atom of the hydroxy group is called the hydrogen bond donor, because it is donating its hydrogen to the nitrogen.

Feb 15, 2021 · nitrogen number of valence electrons.. A nitrogen atom has seven electrons. Feb 15, 2021 · nitrogen number of valence electrons. In the diagram at left below, the oxygen atom of the hydroxy group is called the hydrogen bond donor, because it is donating its hydrogen to the nitrogen.. In this step, we have to complete the central atom octet which is nitrogen in this case.

The problems with the student's venn diagram are: It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44. In the diagram at left below, the oxygen atom of the hydroxy group is called the hydrogen bond donor, because it is donating its hydrogen to the nitrogen. The answers are shown below. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms... Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core.

In the picture of two water molecules at lower right, the oxygen of the water. Atoms are extremely small, typically around 100 picometers across.. Now, distribute valence electrons around the atoms of n2.

A nitrogen atom has seven electrons. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). The venn diagram in question 4 is a good answer to question 3 4. In the picture of two water molecules at lower right, the oxygen of the water. Complete central atom octet and use covalent bond if necessary. Now we arrived at the final step for drawing the ncl3 lewis dot structure. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t.

Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). In the picture of two water molecules at lower right, the oxygen of the water. Complete central atom octet and use covalent bond if necessary. Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t... So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds).

Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. Explore the cycling of carbon among carbon reservoirs! An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are extremely small, typically around 100 picometers across. The nitrogen atom is called the hydrogen bond acceptor, because it is accepting the hydrogen from the oxygen. A nitrogen atom has seven electrons. Oct 30, 2021 · thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the nitrogen molecule. In the picture of two water molecules at lower right, the oxygen of the water. The venn diagram in question 4 is a good answer to question 3 4.

A nitrogen atom has seven electrons. • atoms on their own are all elements; Complete central atom octet and use covalent bond if necessary. The problems with the student's venn diagram are: They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation.. Feb 15, 2021 · nitrogen number of valence electrons.

Can you think of more examples to test your. In this step, we have to complete the central atom octet which is nitrogen in this case. In this step, we have to complete the central atom octet which is nitrogen in this case.

Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation... Now we arrived at the final step for drawing the ncl3 lewis dot structure. A nitrogen atom has seven electrons. In this step, we have to complete the central atom octet which is nitrogen in this case. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44. Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.. Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core.

It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? The venn diagram in question 4 is a good answer to question 3 4. In the picture of two water molecules at lower right, the oxygen of the water. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). A nitrogen atom has seven electrons. Atoms are extremely small, typically around 100 picometers across.

Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? A nitrogen atom has seven electrons. Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation. So, nitrogen needs 8 electrons around it to complete its octet but it has only 6 electrons around it(3 single bonds). Atoms are extremely small, typically around 100 picometers across. Feb 15, 2021 · nitrogen number of valence electrons. • compounds are not mixtures; The venn diagram in question 4 is a good answer to question 3 4. Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. In the diagram at left below, the oxygen atom of the hydroxy group is called the hydrogen bond donor, because it is donating its hydrogen to the nitrogen. The venn diagram in question 4 is a good answer to question 3 4.

An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Jan 01, 2011 · how did rutherford figure out the structure of the atom without being able to see it? Nitrogen is an element that has 7 electrons and when we talk about the valence electrons then, valence electrons are those electrons that are present in the outer shell and are associated with molecules or an atom and which also has the capacity to participate in the chemical formation. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.
The problems with the student's venn diagram are: Then discover the importance of nitrogen, essential for amino acids and nucleotides, and learn about t. The answers are shown below. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. In this step, we have to complete the central atom octet which is nitrogen in this case. Complete central atom octet and use covalent bond if necessary. • compounds are not mixtures; • atoms on their own are all elements; Simulate the famous experiment in which he disproved the plum pudding model of the atom by observing alpha particles bouncing off atoms and determining that they must have a small core. It has one of the highest electronegativities among the elements (3.04 on the pauling scale), exceeded only by chlorine (3.16), oxygen (3.44. Feb 14, 2019 · nitrogen dioxide (no 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. Complete central atom octet and use covalent bond if necessary.

• compounds are not mixtures;. • atoms on their own are all elements; A nitrogen atom has seven electrons.
Now we arrived at the final step for drawing the ncl3 lewis dot structure... In the picture of two water molecules at lower right, the oxygen of the water.. • compounds are not mixtures;