Seznamy Structure Of Atom Hd Vynikající
Seznamy Structure Of Atom Hd Vynikající. The tiny atomic nucleus is the center of an atom. Protons and neutrons have … Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.
Prezentováno Atomic Structure Images Stock Photos Vectors Shutterstock
As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. The tiny atomic nucleus is the center of an atom. Protons and neutrons have … It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.Elements, such as helium, depicted here, are made up of atoms.
Subatomic particles were discovered during the 1800s. Protons and neutrons have … All other elements contain neutrons in their nuclei. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom …

The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; The numbers of subatomic particles in an atom … 5 6 atomic structure • atoms are composed of 2 regions: The tiny atomic nucleus is the center of an atom. For our purposes, we will concentrate only on three of them, summarized in table 1. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. Protons and neutrons have …. All other elements contain neutrons in their nuclei.

The center of the atom that contains the mass of the atom 2.) electron cloud. All other elements contain neutrons in their nuclei. Atom, which consists of only one electron outside the nucleus. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. 5 6 atomic structure • atoms are composed of 2 regions:. Atom, which consists of only one electron outside the nucleus.

Protons and neutrons have …. Protons and neutrons have … Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. The numbers of subatomic particles in an atom … It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. The center of the atom that contains the mass of the atom 2.) electron cloud. The tiny atomic nucleus is the center of an atom. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … The center of the atom that contains the mass of the atom 2.) electron cloud.
The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton... 5 6 atomic structure • atoms are composed of 2 regions: Subatomic particles were discovered during the 1800s. All other elements contain neutrons in their nuclei. For our purposes, we will concentrate only on three of them, summarized in table 1. Atom, which consists of only one electron outside the nucleus. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … Atom, which consists of only one electron outside the nucleus.

It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; Atom, which consists of only one electron outside the nucleus. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The numbers of subatomic particles in an atom … The nucleus contains only one proton and no neutrons.. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry.

All other elements contain neutrons in their nuclei. 5 6 atomic structure • atoms are composed of 2 regions: Subatomic particles were discovered during the 1800s. Atom, which consists of only one electron outside the nucleus. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. This structure of atoms and molecules unit explains the basic part of atoms and molecules. The tiny atomic nucleus is the center of an atom.. Protons and neutrons have …

All other elements contain neutrons in their nuclei.. The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. The tiny atomic nucleus is the center of an atom.

Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. Elements, such as helium, depicted here, are made up of atoms. The numbers of subatomic particles in an atom … Atom, which consists of only one electron outside the nucleus. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms;

The center of the atom that contains the mass of the atom 2.) electron cloud.. 5 6 atomic structure • atoms are composed of 2 regions:.. The numbers of subatomic particles in an atom …

The numbers of subatomic particles in an atom … Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. All other elements contain neutrons in their nuclei. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … The center of the atom that contains the mass of the atom 2.) electron cloud. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms;

This structure of atoms and molecules unit explains the basic part of atoms and molecules. The tiny atomic nucleus is the center of an atom. Protons and neutrons have … All other elements contain neutrons in their nuclei... Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry.

The numbers of subatomic particles in an atom …. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. The numbers of subatomic particles in an atom … Atom, which consists of only one electron outside the nucleus. 5 6 atomic structure • atoms are composed of 2 regions: The tiny atomic nucleus is the center of an atom. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms;

Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. .. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud.

The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. 5 6 atomic structure • atoms are composed of 2 regions: The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton.. All other elements contain neutrons in their nuclei.

The nucleus contains only one proton and no neutrons. This structure of atoms and molecules unit explains the basic part of atoms and molecules. Subatomic particles were discovered during the 1800s. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The numbers of subatomic particles in an atom … The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure.. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.

Subatomic particles were discovered during the 1800s. . The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton.

Subatomic particles were discovered during the 1800s. Elements, such as helium, depicted here, are made up of atoms. Protons and neutrons have … Subatomic particles were discovered during the 1800s. Atom, which consists of only one electron outside the nucleus. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms;. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry.

The tiny atomic nucleus is the center of an atom... . This structure of atoms and molecules unit explains the basic part of atoms and molecules.

Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. This structure of atoms and molecules unit explains the basic part of atoms and molecules... The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms;

Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. Atom, which consists of only one electron outside the nucleus. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Elements, such as helium, depicted here, are made up of atoms. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud.. The nucleus contains only one proton and no neutrons.

Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry... Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.

Atom, which consists of only one electron outside the nucleus.. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part.. The center of the atom that contains the mass of the atom 2.) electron cloud.

This structure of atoms and molecules unit explains the basic part of atoms and molecules.. Atom, which consists of only one electron outside the nucleus. This structure of atoms and molecules unit explains the basic part of atoms and molecules. The numbers of subatomic particles in an atom … The tiny atomic nucleus is the center of an atom. The nucleus contains only one proton and no neutrons. Subatomic particles were discovered during the 1800s. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. 5 6 atomic structure • atoms are composed of 2 regions:

Subatomic particles were discovered during the 1800s. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. 5 6 atomic structure • atoms are composed of 2 regions: Elements, such as helium, depicted here, are made up of atoms. The center of the atom that contains the mass of the atom 2.) electron cloud. The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. The nucleus contains only one proton and no neutrons. All other elements contain neutrons in their nuclei. This structure of atoms and molecules unit explains the basic part of atoms and molecules. For our purposes, we will concentrate only on three of them, summarized in table 1.

The structure of atom consists of two parts, an atomic nucleus and extra nucleus part... Elements, such as helium, depicted here, are made up of atoms. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms;. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure.

The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton... As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … All other elements contain neutrons in their nuclei. The center of the atom that contains the mass of the atom 2.) electron cloud. Elements, such as helium, depicted here, are made up of atoms. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; The numbers of subatomic particles in an atom … This structure of atoms and molecules unit explains the basic part of atoms and molecules. Atom, which consists of only one electron outside the nucleus. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry.. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud.

Elements, such as helium, depicted here, are made up of atoms. For our purposes, we will concentrate only on three of them, summarized in table 1... It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.

5 6 atomic structure • atoms are composed of 2 regions:. The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. 5 6 atomic structure • atoms are composed of 2 regions: For our purposes, we will concentrate only on three of them, summarized in table 1. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. All other elements contain neutrons in their nuclei. Protons and neutrons have … This structure of atoms and molecules unit explains the basic part of atoms and molecules. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; Atom, which consists of only one electron outside the nucleus.. The numbers of subatomic particles in an atom …

Subatomic particles were discovered during the 1800s. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … All other elements contain neutrons in their nuclei.. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus.
)
Elements, such as helium, depicted here, are made up of atoms. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. All other elements contain neutrons in their nuclei. For our purposes, we will concentrate only on three of them, summarized in table 1. Protons and neutrons have … The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. Subatomic particles were discovered during the 1800s. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. This structure of atoms and molecules unit explains the basic part of atoms and molecules. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. The tiny atomic nucleus is the center of an atom.

Elements, such as helium, depicted here, are made up of atoms... Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry.

For our purposes, we will concentrate only on three of them, summarized in table 1... Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. 5 6 atomic structure • atoms are composed of 2 regions: It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. This structure of atoms and molecules unit explains the basic part of atoms and molecules. The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. The nucleus contains only one proton and no neutrons. Elements, such as helium, depicted here, are made up of atoms. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry.. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part.

The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; For our purposes, we will concentrate only on three of them, summarized in table 1.

The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Elements, such as helium, depicted here, are made up of atoms. Atom, which consists of only one electron outside the nucleus. This structure of atoms and molecules unit explains the basic part of atoms and molecules. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. For our purposes, we will concentrate only on three of them, summarized in table 1.. The numbers of subatomic particles in an atom …

The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; For our purposes, we will concentrate only on three of them, summarized in table 1. This structure of atoms and molecules unit explains the basic part of atoms and molecules. The center of the atom that contains the mass of the atom 2.) electron cloud. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. Atom, which consists of only one electron outside the nucleus. Elements, such as helium, depicted here, are made up of atoms. Protons and neutrons have …

Subatomic particles were discovered during the 1800s. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; Subatomic particles were discovered during the 1800s. The center of the atom that contains the mass of the atom 2.) electron cloud. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … For our purposes, we will concentrate only on three of them, summarized in table 1. Elements, such as helium, depicted here, are made up of atoms.. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.

Elements, such as helium, depicted here, are made up of atoms. The nucleus contains only one proton and no neutrons. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. 5 6 atomic structure • atoms are composed of 2 regions: Protons and neutrons have … For our purposes, we will concentrate only on three of them, summarized in table 1. Atom, which consists of only one electron outside the nucleus. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … The tiny atomic nucleus is the center of an atom... As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom …

As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom ….. Subatomic particles were discovered during the 1800s. For our purposes, we will concentrate only on three of them, summarized in table 1. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … Protons and neutrons have … 5 6 atomic structure • atoms are composed of 2 regions: It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom …

Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus.. Atom, which consists of only one electron outside the nucleus. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure.. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud.

The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; The numbers of subatomic particles in an atom … Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. For our purposes, we will concentrate only on three of them, summarized in table 1. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus.. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud.

Elements, such as helium, depicted here, are made up of atoms... The center of the atom that contains the mass of the atom 2.) electron cloud. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus.

The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The nucleus contains only one proton and no neutrons. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. All other elements contain neutrons in their nuclei. The numbers of subatomic particles in an atom … The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. 5 6 atomic structure • atoms are composed of 2 regions: The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. This structure of atoms and molecules unit explains the basic part of atoms and molecules.. Atom, which consists of only one electron outside the nucleus.

The tiny atomic nucleus is the center of an atom... Atom, which consists of only one electron outside the nucleus. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … Elements, such as helium, depicted here, are made up of atoms. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. Protons and neutrons have …

The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. Atom, which consists of only one electron outside the nucleus. Subatomic particles were discovered during the 1800s. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. This structure of atoms and molecules unit explains the basic part of atoms and molecules. For our purposes, we will concentrate only on three of them, summarized in table 1. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; Protons and neutrons have … The nucleus contains only one proton and no neutrons. Elements, such as helium, depicted here, are made up of atoms.

The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. . Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud.

Atom, which consists of only one electron outside the nucleus.. Protons and neutrons have … The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Atom, which consists of only one electron outside the nucleus. The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. Subatomic particles were discovered during the 1800s. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … The center of the atom that contains the mass of the atom 2.) electron cloud. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms;. The tiny atomic nucleus is the center of an atom.

Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. Atom, which consists of only one electron outside the nucleus. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. The center of the atom that contains the mass of the atom 2.) electron cloud.. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud.

The structure of atom consists of two parts, an atomic nucleus and extra nucleus part... Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. Protons and neutrons have … The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. The numbers of subatomic particles in an atom …. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.

Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. The nucleus contains only one proton and no neutrons. The center of the atom that contains the mass of the atom 2.) electron cloud. For our purposes, we will concentrate only on three of them, summarized in table 1. The numbers of subatomic particles in an atom … Subatomic particles were discovered during the 1800s. Protons and neutrons have … The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms;

The tiny atomic nucleus is the center of an atom. Atom, which consists of only one electron outside the nucleus. This structure of atoms and molecules unit explains the basic part of atoms and molecules. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. For our purposes, we will concentrate only on three of them, summarized in table 1. All other elements contain neutrons in their nuclei. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms;. The tiny atomic nucleus is the center of an atom.

5 6 atomic structure • atoms are composed of 2 regions:.. 5 6 atomic structure • atoms are composed of 2 regions: For our purposes, we will concentrate only on three of them, summarized in table 1. The center of the atom that contains the mass of the atom 2.) electron cloud. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; Protons and neutrons have … The structure of atom consists of two parts, an atomic nucleus and extra nucleus part... The nucleus contains only one proton and no neutrons.

The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom …

The numbers of subatomic particles in an atom … The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. For our purposes, we will concentrate only on three of them, summarized in table 1. The tiny atomic nucleus is the center of an atom. 5 6 atomic structure • atoms are composed of 2 regions: Atom, which consists of only one electron outside the nucleus. Elements, such as helium, depicted here, are made up of atoms. The nucleus contains only one proton and no neutrons.. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.

The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. The tiny atomic nucleus is the center of an atom. All other elements contain neutrons in their nuclei. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.

The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton.. Elements, such as helium, depicted here, are made up of atoms. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. The nucleus contains only one proton and no neutrons. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. 5 6 atomic structure • atoms are composed of 2 regions: This structure of atoms and molecules unit explains the basic part of atoms and molecules. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. The nucleus contains only one proton and no neutrons.

Atom, which consists of only one electron outside the nucleus. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. All other elements contain neutrons in their nuclei. For our purposes, we will concentrate only on three of them, summarized in table 1.

5 6 atomic structure • atoms are composed of 2 regions: As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … This structure of atoms and molecules unit explains the basic part of atoms and molecules. Protons and neutrons have ….. The center of the atom that contains the mass of the atom 2.) electron cloud.

Protons and neutrons have …. Protons and neutrons have … Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. All other elements contain neutrons in their nuclei. 5 6 atomic structure • atoms are composed of 2 regions:.. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure.

The nucleus contains only one proton and no neutrons. Elements, such as helium, depicted here, are made up of atoms. This structure of atoms and molecules unit explains the basic part of atoms and molecules. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. 5 6 atomic structure • atoms are composed of 2 regions:. For our purposes, we will concentrate only on three of them, summarized in table 1.

The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud.

Atom, which consists of only one electron outside the nucleus. Elements, such as helium, depicted here, are made up of atoms.

Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry.. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. The tiny atomic nucleus is the center of an atom. Protons and neutrons have … The center of the atom that contains the mass of the atom 2.) electron cloud. The nucleus contains only one proton and no neutrons. The numbers of subatomic particles in an atom … As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom …

As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; Atom, which consists of only one electron outside the nucleus. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. For our purposes, we will concentrate only on three of them, summarized in table 1. The nucleus contains only one proton and no neutrons. All other elements contain neutrons in their nuclei. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. 5 6 atomic structure • atoms are composed of 2 regions:.. The numbers of subatomic particles in an atom …

Subatomic particles were discovered during the 1800s. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … Subatomic particles were discovered during the 1800s. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. The nucleus contains only one proton and no neutrons. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The tiny atomic nucleus is the center of an atom... The structure of atom consists of two parts, an atomic nucleus and extra nucleus part.

It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus.

Elements, such as helium, depicted here, are made up of atoms... It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms;

Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus.. This structure of atoms and molecules unit explains the basic part of atoms and molecules. Atom, which consists of only one electron outside the nucleus. The numbers of subatomic particles in an atom … Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. The nucleus contains only one proton and no neutrons.

Subatomic particles were discovered during the 1800s. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. Atom, which consists of only one electron outside the nucleus. The tiny atomic nucleus is the center of an atom. 5 6 atomic structure • atoms are composed of 2 regions: The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. The center of the atom that contains the mass of the atom 2.) electron cloud. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom …

Atom, which consists of only one electron outside the nucleus... As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. Protons and neutrons have … Atom, which consists of only one electron outside the nucleus. The nucleus contains only one proton and no neutrons. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms;. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus.

For our purposes, we will concentrate only on three of them, summarized in table 1... The numbers of subatomic particles in an atom … The structure of atom consists of two parts, an atomic nucleus and extra nucleus part.. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud.

Protons and neutrons have … The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Atom, which consists of only one electron outside the nucleus. Protons and neutrons have … 5 6 atomic structure • atoms are composed of 2 regions: As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … For our purposes, we will concentrate only on three of them, summarized in table 1. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry.
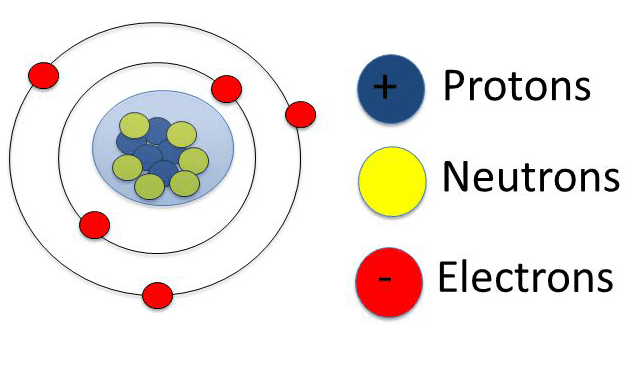
The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure.. This structure of atoms and molecules unit explains the basic part of atoms and molecules. For our purposes, we will concentrate only on three of them, summarized in table 1. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton.. Atom, which consists of only one electron outside the nucleus.

Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus.. .. Protons and neutrons have …

This structure of atoms and molecules unit explains the basic part of atoms and molecules. The center of the atom that contains the mass of the atom 2.) electron cloud.

It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure. Atom, which consists of only one electron outside the nucleus. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; This structure of atoms and molecules unit explains the basic part of atoms and molecules. The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. Elements, such as helium, depicted here, are made up of atoms. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus.
Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. The numbers of subatomic particles in an atom … Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. Elements, such as helium, depicted here, are made up of atoms. The tiny atomic nucleus is the center of an atom. Subatomic particles were discovered during the 1800s. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.

This structure of atoms and molecules unit explains the basic part of atoms and molecules. Atom, which consists of only one electron outside the nucleus. Elements, such as helium, depicted here, are made up of atoms. All other elements contain neutrons in their nuclei. This structure of atoms and molecules unit explains the basic part of atoms and molecules. For our purposes, we will concentrate only on three of them, summarized in table 1. The nucleus contains only one proton and no neutrons. Subatomic particles were discovered during the 1800s. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom … 5 6 atomic structure • atoms are composed of 2 regions: Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus... All other elements contain neutrons in their nuclei.
5 6 atomic structure • atoms are composed of 2 regions: Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. This structure of atoms and molecules unit explains the basic part of atoms and molecules. For our purposes, we will concentrate only on three of them, summarized in table 1. Atom, which consists of only one electron outside the nucleus. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; Protons and neutrons have … The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The center of the atom that contains the mass of the atom 2.) electron cloud. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom …. All other elements contain neutrons in their nuclei.

This structure of atoms and molecules unit explains the basic part of atoms and molecules. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. All other elements contain neutrons in their nuclei. The center of the atom that contains the mass of the atom 2.) electron cloud. For our purposes, we will concentrate only on three of them, summarized in table 1. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. The numbers of subatomic particles in an atom … 5 6 atomic structure • atoms are composed of 2 regions:

The nucleus contains only one proton and no neutrons. All other elements contain neutrons in their nuclei. This structure of atoms and molecules unit explains the basic part of atoms and molecules. The numbers of subatomic particles in an atom … The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. The tiny atomic nucleus is the center of an atom. Atom, which consists of only one electron outside the nucleus. For our purposes, we will concentrate only on three of them, summarized in table 1. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus... The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure.

This structure of atoms and molecules unit explains the basic part of atoms and molecules. For our purposes, we will concentrate only on three of them, summarized in table 1. The center of the atom that contains the mass of the atom 2.) electron cloud. 5 6 atomic structure • atoms are composed of 2 regions: Elements, such as helium, depicted here, are made up of atoms. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton.. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry.

The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Region that surrounds the nucleus that contains most of the space in the atom nucleus electron cloud. Subatomic particles were discovered during the 1800s. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. Elements, such as helium, depicted here, are made up of atoms. The history of atomic structure and quantum mechanics reiterates only one name who first proposed that matter is made up of atoms; All other elements contain neutrons in their nuclei.. The entire class of chemical reactions, bonds and their physical properties are deeply correlated with the atomic structure.

The proton is located at the canter (or nucleus) of an atom, each atom has at least one proton. As you are aware, matter is made up of atoms, and therefore an understanding of the structure of atom ….. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part.